 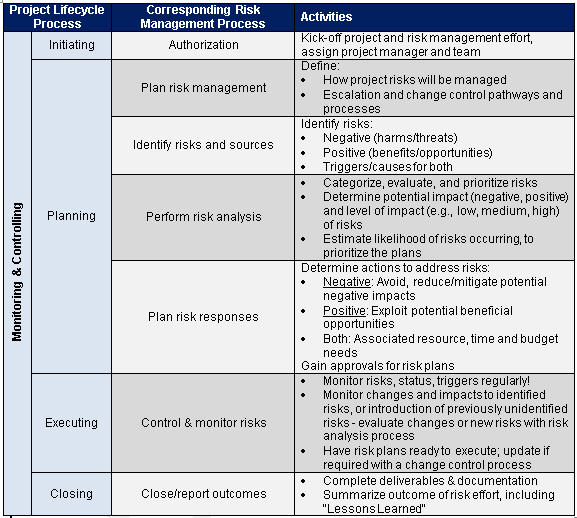
Through examples it shares practical applications implementing tools described by several of the recently enacted or updated standards and technical reports relevant and applicable to medical device risk management, (ISO/EN 14971:2012 with a 2019 update summary (little change in Risk Management process), what will be in the TR/ISO 24971:2013->2019, risk as related to 21 CFR 820 and as related to ISO 13485:2016, impacts of software risk assessment for IEC 62304:2015, and impacts of risk on human factors engineering IEC 62366-1:2015). This course illustrates commonly used risk-identification and risk-reducing methods. Using case studies and interaction, you will practice identifying and analyzing potential product and process hazards, FMEA, hazard and Fault Tree Analysis, Hazard and Critical Control Point, and all the critical skills needed to create a risk management plan, process, report and file. 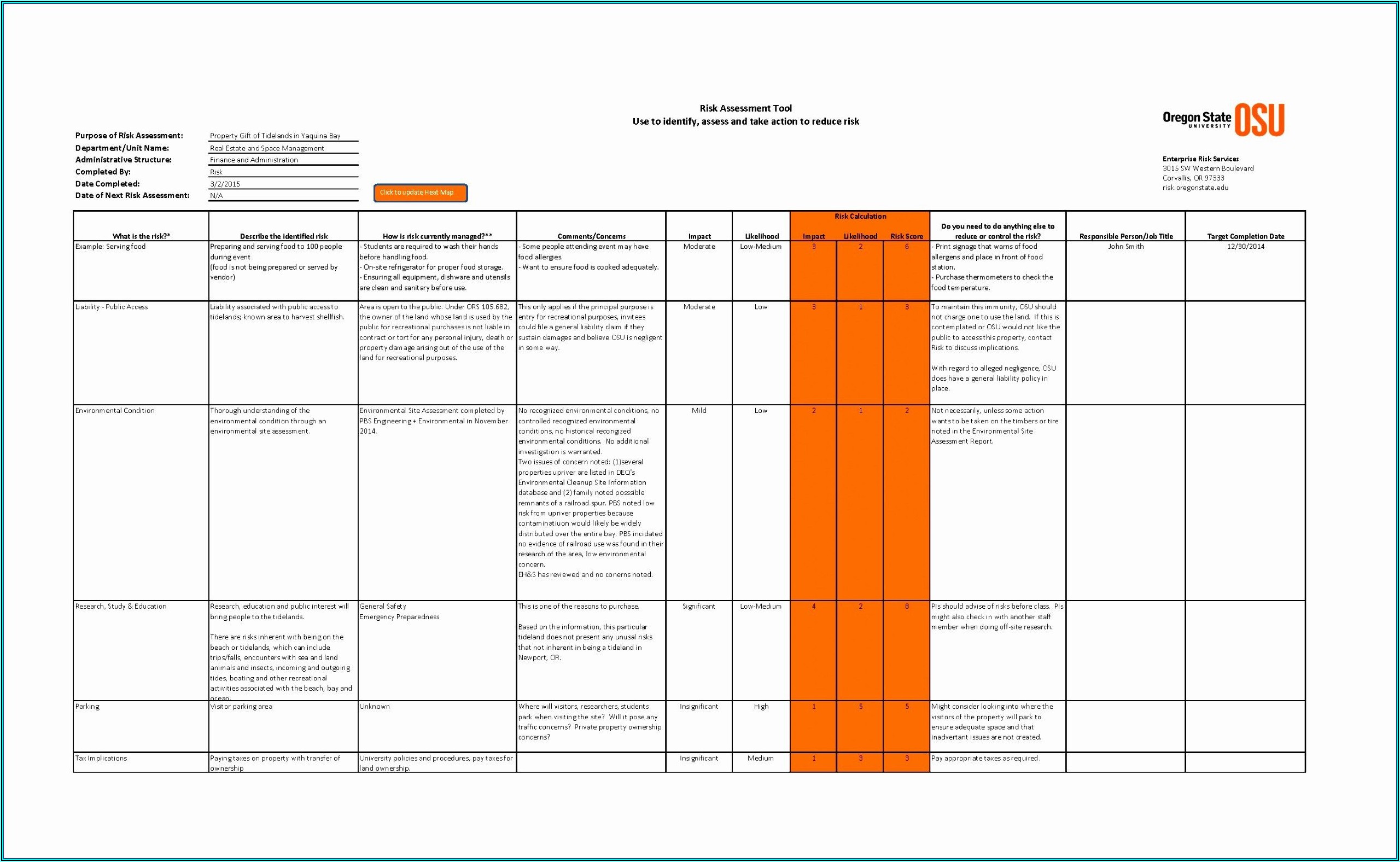 
It also focuses on recently enacted standards specifically related to medical device risk management. Designed for engineers, technicians, and professionals focusing on product and process risk, this course teaches you the common risk-management methods used in product design and manufacturing processes. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
